In heat exchanger design, one idea appears very logical:
If a metal conducts heat well, the exchanger should perform better.
And in theory, this is true.
Metals like:
- copper
- aluminum
- some special alloys
have very high thermal conductivity. They allow heat to pass through quickly, with very little resistance.
So naturally, one might expect that using high-conductivity metals should always give superior thermal performance.
But in real process plants, this is not always what happens.
Exchangers built with excellent heat-conducting materials can still:
- miss duty
- foul quickly
- lose performance over time
- require frequent maintenance
This article explains why high-conductivity metals alone cannot guarantee good performance, and why plant reality often overrides theoretical advantage.
Table of Contents
Conduction Is Only One Part of the Heat Path
Heat transfer through an exchanger involves multiple steps:
- heat moving from hot fluid to metal surface
- heat passing through the metal wall
- heat entering the cold fluid
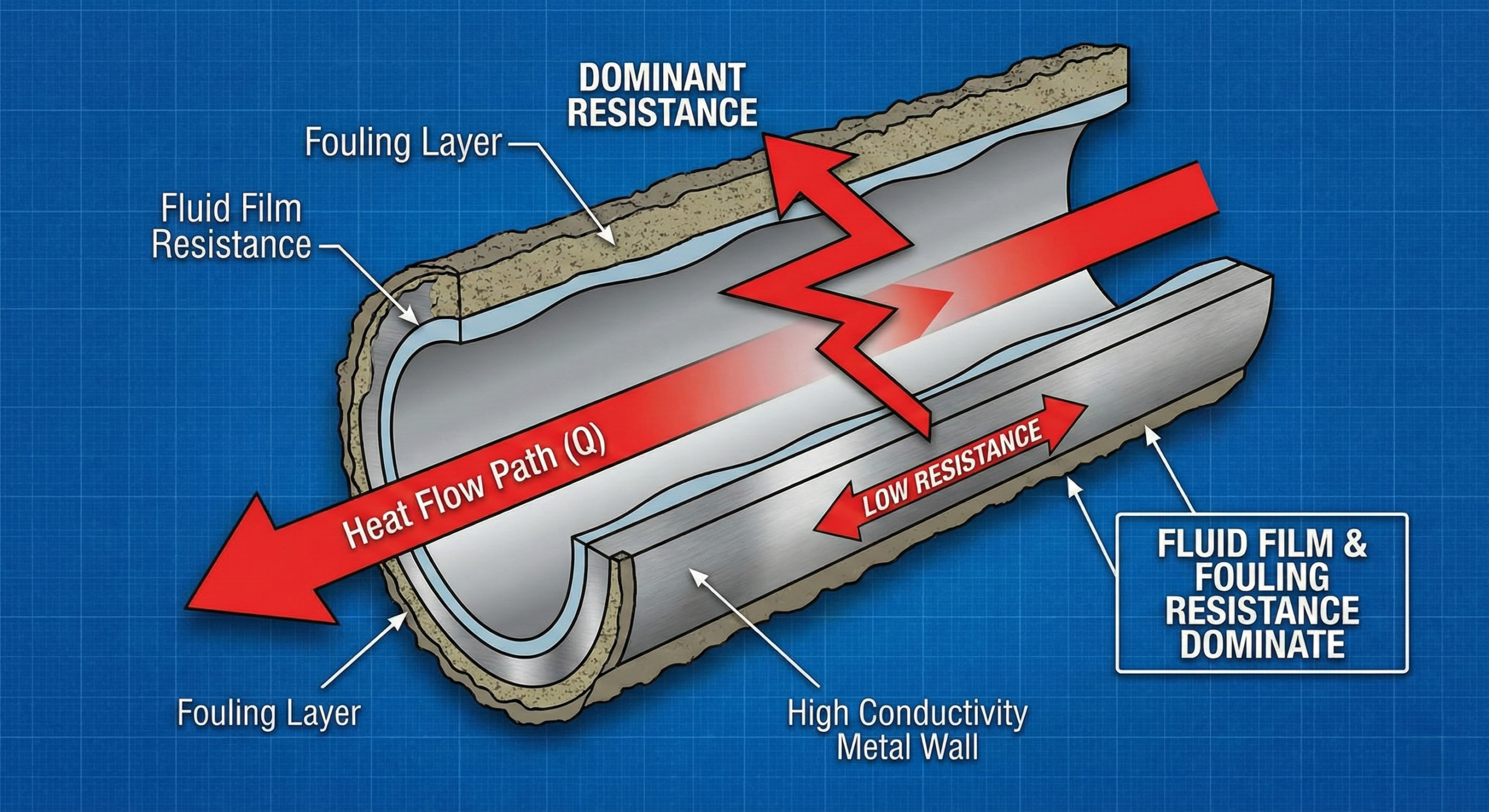
The metal wall is only one part of this journey.
Even if the wall conducts heat extremely well, other resistances may dominate:
- fluid film resistance
- fouling layers
- flow maldistribution
If those resistances are high, improving the metal does not change the outcome much.
Fluid-Side Resistance Often Dominates
In many process services:
- oil is viscous
- gas has poor heat transfer properties
- flow may be laminar
In such cases:
- the main limitation is the fluid film near the wall
- the metal wall contributes only a small portion of total resistance
So switching from steel to a higher-conductivity metal may produce only a small improvement.
The bottleneck remains in the fluid, not the wall.
Fouling Quickly Cancels Material Advantage
Even the best metal surface becomes less effective once deposits form.
Fouling layers:
- act like insulation
- increase thermal resistance
- block direct heat transfer
After fouling develops:
- heat must pass through the deposit first
- then through the metal
So the difference between metals becomes less important.
A thin fouling layer can have more resistance than the metal itself.
This is why an exchanger may perform very well when new, but slowly lose the benefit of high-conductivity materials as fouling builds.
Corrosion Can Damage Good Conductors
Some high-conductivity metals are more sensitive to certain environments.
For example:
- copper can suffer in corrosive fluids
- aluminum can degrade in some chemical services
Corrosion can cause:
- surface roughness
- pitting
- thinning
- deposit attachment
These effects reduce effective heat transfer over time.
So even if the metal was thermally excellent at the start, service conditions may slowly damage its performance.
Mechanical Strength Is Often Lower
High-conductivity metals sometimes have lower mechanical strength compared to steels.
This can lead to:
- deformation under pressure
- vibration damage
- fatigue over long service
To compensate, designers may:
- increase wall thickness
- add structural support
But increasing thickness increases conduction resistance.
So the thermal advantage may be partially offset by mechanical requirements.
Compatibility with Process Fluids Matters
Thermal conductivity is only one factor in material selection.
Materials must also be compatible with:
- process chemistry
- temperature range
- corrosion conditions
- cleaning methods
Sometimes a lower-conductivity metal is chosen because:
- it lasts longer
- it resists corrosion better
- it requires less maintenance
A slightly lower heat transfer performance may be acceptable if reliability improves.
Fouling Adhesion Can Be Stronger on Some Surfaces
In certain services, deposits stick more easily to some metals than others.
For example:
- rough or reactive surfaces may attract scaling
- certain metals may promote deposit formation
So even if a metal transfers heat well initially, it may foul faster.
Over time:
- the performance advantage disappears
- cleaning frequency increases
Material choice influences not just conduction, but also how the surface behaves in real fluids.
Temperature Distribution Changes with Service Conditions
Even with high-conductivity metal, performance depends on:
- flow rate
- temperature difference
- operating stability
If conditions change:
- fluid-side resistance may increase
- fouling may accelerate
- driving force may reduce
So the metal cannot compensate for poor operating conditions.
Heat transfer is a system effect, not a material effect alone.
High Conductivity Does Not Prevent Heat Loss
In some cases, very conductive metals can even increase heat loss to surroundings if insulation is not adequate.
For example:
- a highly conductive pipe wall can quickly carry heat to the outer surface
- if insulation is weak, heat escapes faster
So high conductivity is beneficial inside the exchanger but must be managed properly in exposed systems.
Real Plant Performance Depends on the Whole System
An exchanger’s performance depends on:
- fluid flow
- turbulence
- fouling behavior
- driving force
- surface condition
- operating history
The metal wall is just one element.
Even the best material cannot overcome poor system conditions.
This is why two exchangers made from different metals may perform similarly in practice.
Why Carbon Steel Still Works Well in Many Services
Carbon steel is not the best thermal conductor.
Yet it is widely used.
Because it offers:
- good strength
- reasonable conductivity
- low cost
- acceptable corrosion resistance in many services
And most importantly:
- fluid-side resistance often dominates anyway.
So the difference between materials may not justify higher cost or complexity.
Owner Perspective: Reliability Often Matters More Than Conductivity
From a plant management point of view, the best material is not always the one with the highest conductivity.
It is the one that gives:
- stable performance
- long life
- low maintenance
- predictable behavior
If a high-conductivity metal:
- corrodes faster,
- fouls quickly,
- or requires frequent replacement,
its thermal advantage may not translate into overall value.
A Simple Way to Understand the Limitation
Think of heat transfer like water flowing through a series of pipes.
If one section is wide and smooth, but another section is narrow and clogged, the flow is controlled by the narrow part.
In heat exchangers:
- metal wall is one section
- fluid films and fouling are others
Even if the wall is excellent, the narrow sections elsewhere still limit the flow of heat.
Final Perspective
High-conductivity metals are valuable tools in heat exchanger design.
They help reduce conduction resistance.
They support efficient heat flow.
But they are not a complete solution.
In real service, performance depends more on:
- fluid behavior
- fouling growth
- operating conditions
- long-term stability
A metal may conduct heat very well in theory.
But in the plant, its success depends on how it survives the environment around it.
Understanding why high-conductivity metals still fail in service helps shift focus from material alone to the full thermal system — where real performance is actually decided.
A practicing chemical engineer with 17+ years of experience in process design, project execution, commissioning, and plant operations. Focused on practical engineering judgment beyond textbook explanations.
