In process plants, two terms are often used as if they mean the same thing:
- “The system is at equilibrium.”
- “The plant is in steady state.”
They sound similar.
Both suggest stability.
Both suggest nothing is changing.
But thermodynamically, they describe completely different conditions.
This confusion may seem small, but it leads to major misunderstandings in:
- heat transfer behavior
- process control interpretation
- troubleshooting decisions
- expectations from equipment performance
This article explains the difference between equilibrium and steady state in simple, plant-oriented terms — and why understanding it changes how people interpret what they see every day.
Table of Contents
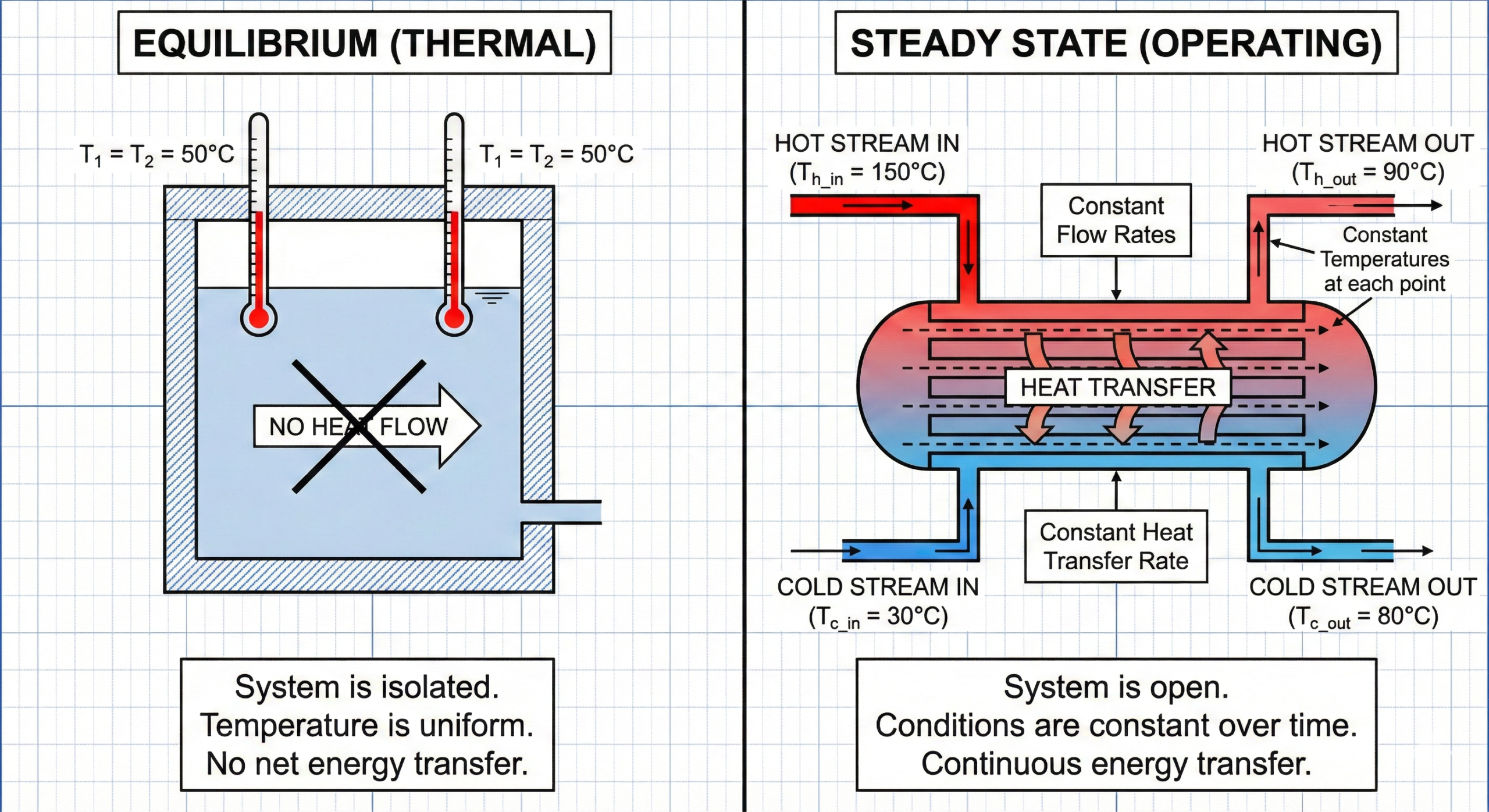
What Is Equilibrium?
Equilibrium is a condition where nothing is trying to change anymore.
In thermal terms, equilibrium means:
- temperatures are equal
- no heat is flowing
- no driving force exists
Nature has reached balance.
There is no movement of energy because there is no reason for movement.
Simple example
Imagine a hot object and a cold object placed together.
- Heat flows from hot to cold.
- Temperatures slowly approach each other.
- Eventually, both reach the same temperature.
At that point:
- heat flow stops,
- temperatures stay constant,
- equilibrium is reached.
No further change occurs.
What Is Steady State?
Steady state is completely different.
Steady state means:
- conditions are not changing with time
- but movement is still happening continuously
In a process plant, steady state means:
- flow is continuous
- heat is continuously moving
- temperatures stay stable
- production continues
So in steady state:
- energy is moving
- mass is moving
- work is being done
But measurements remain constant.
The Key Difference
The simplest way to understand it is:
- Equilibrium = no driving force, no flow
- Steady state = driving force exists, flow continues, but conditions stay constant
This difference is critical for heat transfer understanding.
Because heat transfer only happens when the system is not at equilibrium.
A Heat Exchanger Is Never at Equilibrium
Consider a working heat exchanger.
Hot fluid enters.
Cold fluid enters.
Heat flows from one to the other.
At the outlet:
- temperatures stabilize
- performance looks constant
- readings stop changing
The exchanger is in steady state.
But it is not at equilibrium.
Why?
Because:
- the hot side is still hotter than the cold side
- temperature difference still exists
- heat is still flowing
If equilibrium were reached:
- both fluids would have the same temperature
- heat flow would stop
- the exchanger would stop doing its job
So equilibrium would mean failure.
Why This Confusion Happens in Plants
People often use “equilibrium” to describe a stable condition.
For example:
- “The column has reached equilibrium.”
- “The unit is in equilibrium now.”
What they usually mean is:
- the system has stabilized
- readings are no longer fluctuating
But the correct word is steady state.
The process is stable, but heat and mass transfer are still actively happening.
Heat Transfer Exists Only Away from Equilibrium
This is one of the most important ideas in thermal engineering:
Heat flows only when temperatures are different, which explains why heat flows spontaneously in thermal systems.
That difference is called the driving force for heat transfer.
That difference is called the driving force.
As long as there is a temperature difference:
- heat transfer continues
- energy moves
- equipment performs its function
If equilibrium is reached:
- temperature difference becomes zero
- heat flow stops
So equilibrium is not the goal in most process equipment.
It is the condition where transfer stops.
Why Plants Must Stay Away from Equilibrium
Every major heat transfer process depends on avoiding equilibrium:
- heaters need hot utility to be hotter than process fluid
- coolers need cooling water to be colder than process fluid
- condensers need temperature difference to remove heat
- reboilers need heat flow into the liquid
If temperatures equalize:
- heat transfer stops — even though heat transfer cannot be completely stopped in real systems.
- duty collapses
- process performance fails
So steady operation requires staying far enough from equilibrium.
Steady State Does Not Mean Nothing Is Happening
In steady state:
- heat is flowing continuously
- energy is entering and leaving
- process streams are changing temperature
But because input equals output:
- conditions appear constant
This can be misleading.
Just because temperatures are stable does not mean the system is inactive.
It means:
- transfer is happening at a balanced rate.
Why This Matters in Troubleshooting
When performance starts drifting, people sometimes assume:
- the system is still “balanced”
- conditions look stable
But the real question is:
- has the driving force reduced?
For example:
- cooling water becomes warmer
- temperature difference shrinks
- heat transfer rate drops
The system may still appear steady.
But it is now closer to equilibrium.
And that means lower heat transfer capacity.
Fouling Moves Systems Closer to Equilibrium
Fouling increases thermal resistance.
As resistance grows:
- more temperature difference is needed for the same heat transfer
- outlet temperatures shift
- approach temperatures tighten
In extreme cases:
- process temperature starts approaching utility temperature
- driving force nearly disappears
At that point, the system is moving toward equilibrium.
And performance collapses.
Why Steady State Can Hide Problems
One of the reasons plants miss early thermal issues is because:
- temperatures look stable
- production continues
- control loops hold values steady
But behind the scenes:
- more steam is being used
- cooling demand is rising
- margin is shrinking
The system is still in steady state.
But it is operating closer to thermal limits.
Operators See Stability. Engineers See Distance from Equilibrium.
Two people looking at the same exchanger may interpret it differently.
An operator may see:
- stable temperatures
- steady operation
An engineer may ask:
- how close are the outlet temperatures getting to each other?
- is the driving force shrinking?
- is the system moving toward equilibrium?
This difference in thinking changes how early problems are detected.
Why Equilibrium Is Important in Separation Processes
In some equipment, equilibrium is desirable.
For example:
- in distillation, vapor and liquid try to reach equilibrium at each stage
- in absorbers, components distribute based on equilibrium relationships
But heat transfer is different.
It requires a temperature difference to keep energy moving.
So for heat transfer equipment:
- equilibrium is not the target
- steady state is the goal
Owner Perspective: Stability vs Capability
From an ownership viewpoint, steady operation is not enough.
A plant may look stable, but if it is operating close to equilibrium:
- there is little thermal margin
- small changes can cause instability
- throughput increases become difficult
Understanding the difference helps identify how much performance headroom really exists.
Final Perspective
Equilibrium and steady state are not the same.
Equilibrium means:
- no temperature difference
- no heat flow
- no transfer
Steady state means:
- heat is moving
- conditions are stable
- work is being done continuously
Process plants depend on steady state, not equilibrium.
Because as soon as equilibrium is reached, heat transfer stops — and when heat transfer stops, the process stops.
Understanding this difference changes how people see:
- temperature readings
- exchanger performance
- thermal margins
- early warning signs
It is a simple concept, but once understood, it brings much deeper clarity to how real plants behave.
Explore the complete series in the Heat Transfer Engineering Hub.
A practicing chemical engineer with 17+ years of experience in process design, project execution, commissioning, and plant operations. Focused on practical engineering judgment beyond textbook explanations.
