In process plants, heat does not wait for instruction.
It moves on its own.
This movement is not caused by pumps, controllers, or operator actions. It occurs naturally whenever conditions allow it.
Understanding why heat flows spontaneously is essential to understanding why plants behave the way they do, why certain limits cannot be crossed, and why some problems repeat regardless of design effort.
This article explains that natural tendency clearly and practically, without mathematical language, and with direct relevance to real plant behavior and also explains why heat flows spontaneously in every process plant, and why engineers can only manage heat flow—not stop it.
Table of Contents
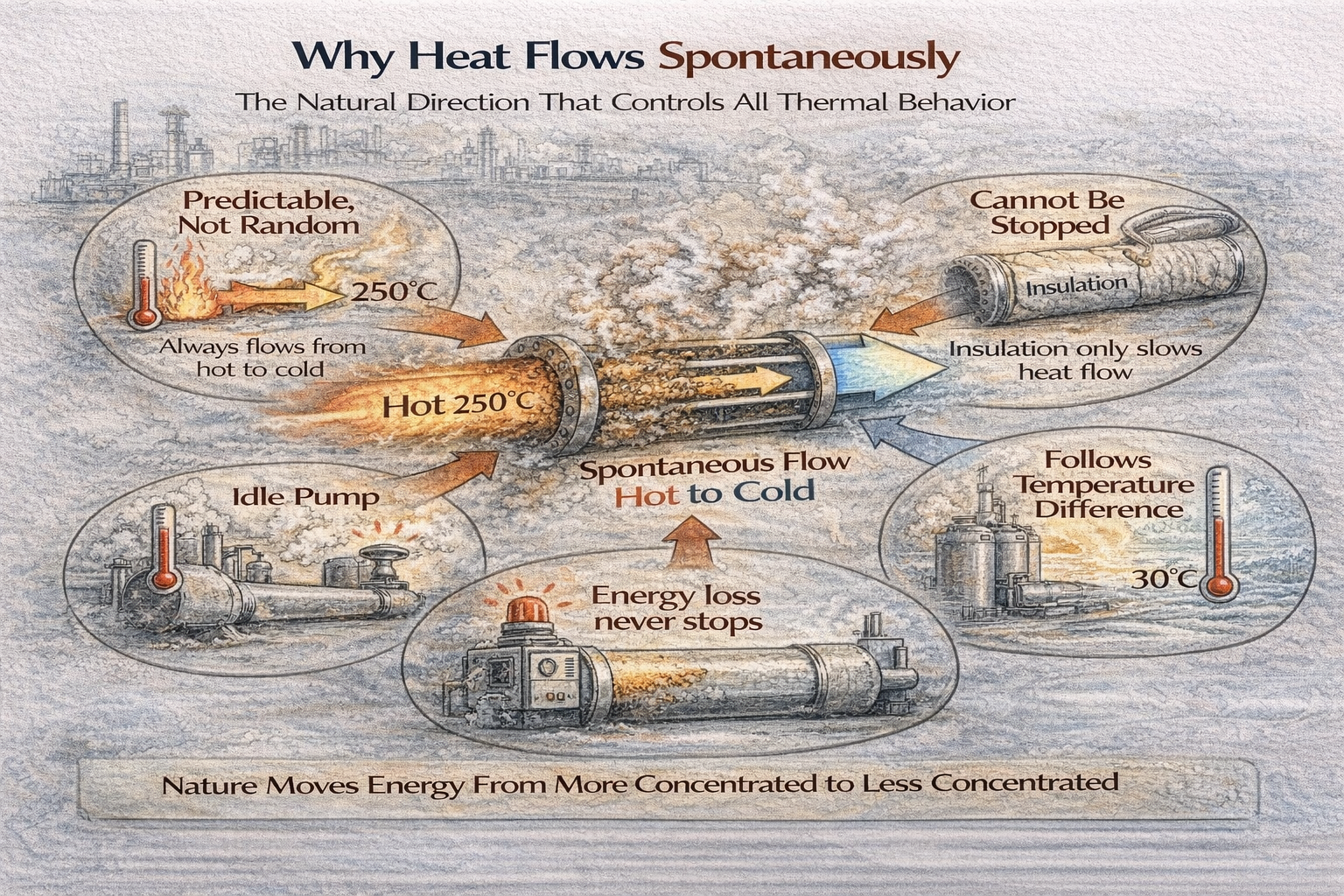
Spontaneous Does Not Mean Random
The word spontaneous is often misunderstood.
Spontaneous heat flow is:
- predictable,
- directional,
- governed by physical law.
It does not mean uncontrolled or chaotic.
Heat flows spontaneously because systems naturally move toward lower energy imbalance. Whenever two regions are at different temperatures, energy redistribution begins immediately.
No trigger is required.
No approval is needed.
Temperature Difference Creates Imbalance
A temperature difference represents an imbalance in energy distribution and becomes the driving force for heat transfer between two regions.
The hotter region contains energy that is more concentrated.
The cooler region contains energy that is less concentrated.
Nature responds by redistributing energy until the imbalance reduces.
This explains why:
- hot fluids cool when exposed to cooler surroundings,
- cold streams warm up when contacted with hot surfaces,
- equipment left idle slowly approaches ambient temperature.
As long as imbalance exists, heat will move.
Why Direction Is Always From Hot to Cold
Heat always flows from higher temperature to lower temperature.
This direction is fixed.
Reversing it requires external energy input, such as:
- steam heating,
- electrical heaters,
- compressors in refrigeration systems.
Without such input, heat cannot move uphill against temperature difference.
This explains why:
- cooling happens naturally,
- heating requires effort,
- insulation only slows heat loss,
- equilibrium is unavoidable.
In plants, expecting heat to move against temperature without energy input leads to flawed design assumptions.
Spontaneous Flow Does Not Mean Fast Flow
Spontaneous heat flow can be:
- rapid,
- gradual,
- barely noticeable.
The rate depends on:
- temperature difference,
- available heat transfer area,
- resistance to heat flow,
- flow conditions.
For example:
- a hot metal surface cools quickly in air,
- a well-insulated vessel cools slowly,
- a large tank retains heat for hours.
Spontaneity explains why heat moves.
Rate explains how fast.
Why Heat Flows Even When Plants Do Not Want It To
Many heat flows in plants are unintended.
Examples include:
- heat loss from insulated piping,
- heat gain in cold storage lines,
- warming of idle equipment,
- cooling of hot standby systems.
These occur not because of poor operation, but because temperature differences exist and paths remain open.
Plants do not choose these heat flows.
They inherit them.
Spontaneous Heat Flow Explains Idle Equipment Behavior
Equipment does not remain thermally static when idle.
A hot system left isolated will:
- lose heat through walls,
- cool through supports,
- exchange energy with surroundings.
A cold system will:
- absorb heat,
- warm up gradually,
- approach ambient temperature.
This behavior surprises operators expecting isolation to mean thermal separation.
Understanding spontaneous heat flow prevents misinterpretation during startups and shutdowns.
Why Insulation Does Not Stop Heat Flow
Insulation does not block heat.
It increases resistance.
As long as:
- temperature difference exists,
- material paths remain,
heat will continue to flow.
This explains why:
- insulated systems still lose energy,
- insulation thickness affects rate, not direction,
- damaged insulation increases losses dramatically.
Spontaneity explains inevitability.
Insulation controls speed.
Spontaneous Heat Flow During Transients
During startups, shutdowns, and disturbances, spontaneous heat flow becomes more visible.
Rapid temperature changes create strong imbalances.
Heat flows:
- into cold equipment,
- out of hot equipment,
- across connected systems.
If not anticipated, this leads to:
- thermal stress,
- delayed stabilization,
- unexpected temperature gradients.
Plants that understand spontaneous heat flow plan transients more safely.
Why Heat Flow Ignores Boundaries Humans Create
Valves, blinds, and isolation procedures stop fluid movement.
They do not stop heat movement.
Heat flows through:
- metal connections,
- structural supports,
- common foundations,
- shared walls.
This is why:
- isolated systems influence each other,
- standby equipment warms unexpectedly,
- cold systems suffer condensation.
Spontaneous heat flow follows physical continuity, not operational boundaries.
Spontaneous Flow Explains Why Energy Loss Is Continuous
Energy loss in plants is not event-based.
It is continuous.
Heat flows out of:
- hot vessels,
- pipelines,
- reactors,
- exchangers.
It flows into:
- refrigerated systems,
- cryogenic lines,
- cooled storage.
Because this flow is spontaneous, losses occur even when plants operate normally.
Reducing losses means managing paths, not eliminating spontaneity.
Why Control Systems Cannot Prevent Spontaneous Heat Flow
Control systems regulate variables.
They do not change physical laws.
A controller can:
- adjust utility flow,
- maintain temperature setpoints.
It cannot:
- reverse natural heat flow,
- stop conduction through metal,
- prevent ambient interaction, because heat transfer cannot be completely stopped.
This explains why:
- perfect control tuning cannot eliminate heat loss,
- controllers work harder as resistance increases,
- control stability degrades when thermal paths change.
Owner Perspective: Spontaneous Heat Flow and Cost
From an owner’s perspective, spontaneous heat flow:
- consumes energy continuously,
- increases operating cost,
- reduces efficiency silently.
Because losses are gradual and distributed, they are often underestimated.
Investments in:
- insulation,
- heat recovery,
- thermal isolation,
do not stop heat flow.
They slow it enough to make operation economical.
Final Perspective
Heat does not wait to be moved.
It moves because imbalance exists.
This spontaneous movement governs how plants heat up, cool down, stabilize, and degrade over time.
Recognizing this tendency removes false expectations and improves judgment.
It is not an advanced concept.
It is a basic reality.
If heat flows naturally whenever temperatures differ, the next question is what determines how fast heat actually moves inside process equipment.
In real plants, the rate of heat transfer depends on temperature difference, thermal resistance, and fluid behavior in equipment.
Driving Force for Heat Transfer Explained
This article explains the concept of driving force and how temperature differences translate into real heat transfer rates inside process plants.
Explore the complete series in the Heat Transfer Engineering Hub.
A practicing chemical engineer with 17+ years of experience in process design, project execution, commissioning, and plant operations. Focused on practical engineering judgment beyond textbook explanations.
